Members will most likely have noticed that AIM has been making revisions to food product labels. Such changes are for important, time-sensitive reasons.
Back in May 2016, the U.S. Food and Drug Administration (FDA) made an announcement that the rules regarding Nutrition Facts on food products would have to change to reflect the latest scientific information.
The most notable new info is the link between diet and chronic diseases that The AIM Companies™ has recognized since 1982, when the incomparably nutritious juice powder of barley grass was first introduced.
One of the FDA’s main reasons for this “refreshed design” in labeling is to make it easier for consumers to make informed decisions about their food choices, particularly in regard to servings, calories and sugar.

Bigger and Bolder Changes
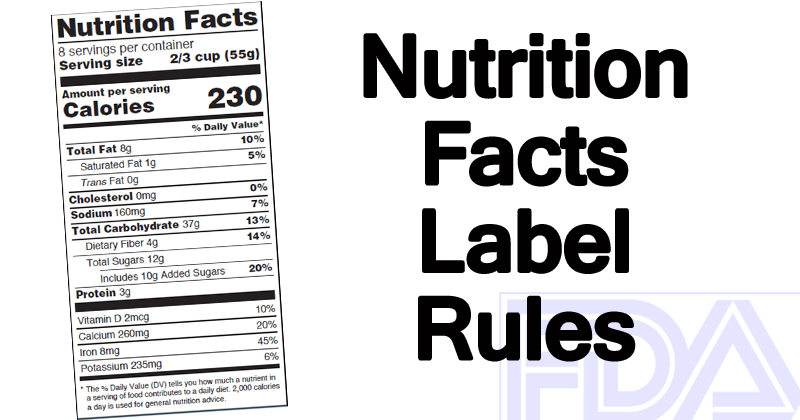
Figuring out what the information on a label means hasn’t always been easy. With the new format, important details are much easier to read:
- Right at the top under Nutrition Facts, the servings per container and the number of serving sizes are bigger and bolder.
- The biggest and boldest detail of them all is calories per serving, one of the most important bits of information when it comes to weight management.
- The addition of added sugars shows the huge difference between what is naturally sweet and unnaturally sweetened.
And based on new scientific evidence, updated daily values for nutrients have been added as well. Speaking of % Daily Values, there is now a clearer definition of what it actually means: how much a nutrient in a serving of food contributes to a daily diet.
The Deadline for Change
The original due date for completing the design of these new labels was July 28, 2018. After the rules were finalized, industry and consumer groups provided feedback regarding the need for more time to make the label changes. In October 2017, the FDA published a proposal to extend the compliance date by approximately one and a half years.
AIM is already ahead of the game, having implemented changes to some of our product labels. More labels will be revised to reflect the new rules. At the same time, the additional costs involved with such a monumental change will be minimized. For example, labels with the former Nutrition Facts can now be used past the old deadline of July 2018, complying with the FDA’s acknowledgment that consumers will see both old and the new versions of labels during the transition period.
So keep your eye on labels!
-◊◊◊-
The AIM Companies has been in business since 1982 providing whole-food concentrates and nutritional supplements through network marketing.